The decade of the “promising pilot” is over. In 2026, the narrative of AI in African healthcare has shifted from aspirational headlines to verifiable, population-level impact. As the continent faces a shortfall of 6 million healthcare workers, artificial intelligence is no longer a luxury—it is the primary “leapfrog” technology closing the gap between 11% of the world’s population and 24% of its disease burden.
From Ethiopia’s national AI-powered TB screening rollout to the 1.8 million obstetric scans completed across Kenya and South Africa, the data is finally in. However, the transition to scale has revealed a new set of “2026 challenges”: the rise of African data sovereignty, the infrastructure “ceiling” of rural connectivity, and the critical safety nuances of LLM clinical decision support.
This report examines the tools currently live at scale, the landmark Penda Health/OpenAI study findings, and the emerging African-built AI ecosystem. We analyze the evidence from Q1 2026 to separate the hype from the high-yield deployments that are fundamentally reshaping patient outcomes in sub-Saharan Africa.
Beyond the pilots — diagnostic AI, maternal health tools, supply chain intelligence, and the evidence that actually exists
| ~6M | Healthcare worker shortfall in sub-Saharan Africa — the structural gap AI is being deployed to bridge |
| 55M+ | People screened by CAD4TB in 90+ countries — the world’s most validated AI TB screening tool |
| 1.8M | Obstetric ultrasound scans completed across Kenya & South Africa by July 2025 (Butterfly Network) |
| 39,849 | Patient visits in landmark Nairobi AI clinical decision support study (Penda Health / OpenAI, 2025) |
| 225 | AI-powered X-ray machines distributed nationally across all regions of Ethiopia for TB screening |
| $150M | US State Department expansion deal for Zipline AI drone delivery across 5 African countries (Nov 2025) |
| $50M | Gates Foundation / OpenAI Horizon1000 commitment — 1,000 primary care clinics with AI by 2028 |
A healthcare worker in a rural Kenyan clinic using an AI-powered diagnostic tool on a tablet to analyze patient X-rays in real-time.
Why 2026 Is Different
For most of the past decade, articles about AI in African healthcare looked the same: promising pilots, exciting potential, a few thousand patients, and then silence. The 2026 landscape is meaningfully different. A small but growing number of tools have crossed the threshold from ‘interesting experiment’ to ‘verifiable deployment at scale with outcome data.’
That shift does not mean the hype has ended — it hasn’t. Many tools still sit in the pilot gap, and several high-profile 2026 announcements describe ambitions rather than current realities. Nevertheless, the evidence base is now real enough to examine critically.
While the broader rise of AI in Africa has touched every sector from fintech to agriculture, 2026 marks the year the evidence finally caught up with the claims in the medical field.
Pilot era vs. 2026 reality: at a glance
| Pilot Era (pre-2024) | 2026 Reality |
| Mostly small pilots in 2–3 countries | Documented deployments in 13+ countries |
| CAD4TB tested on thousands of patients | CAD4TB has screened 55M+ people globally; Ethiopia alone has 225 national units |
| Obstetric ultrasound at a handful of pilot sites | 1.8M scans in 697+ Kenya & South Africa facilities (Butterfly Network) |
| LLM decision support = theory or benchmarks | 40,000-patient live RWE from Nairobi primary care (Penda/OpenAI, 2025) |
| Supply chain AI = donor-funded pilots | Zipline: delivery every 60 seconds, 4,800+ facilities, $150M US expansion deal |
| No national AI health programmes | Ethiopia: 225 AI X-ray units nationally; Rwanda: National Health Intelligence Centre |
| Regulation: almost entirely absent | Kenya: National AI Strategy 2025–2030; Data localisation laws in Kenya, Nigeria, Ghana |
The table above is the article’s central argument. Every cell in the right column is documented below with sources, scale indicators, and — where they exist — outcome data.
The structural deficit that makes AI necessary
The numbers that follow are the permanent context for every deployment discussed in this piece. They explain why tools are being built and why the ambition outpaces the evidence.
| 11% / 24% | Africa holds 11% of the world’s population but 24% of the global disease burden |
| 3% / 1% | Yet the continent has only 3% of the world’s health workers and less than 1% of global health expenditure |
| 1:500,000 | Nigeria’s pathologist-to-population ratio vs. the global average of 1:25,000 |
| 6–8M | Deaths annually in LMICs attributable to low-quality care, per WHO estimates |
| 180 yrs | How long Rwanda would take to close its health worker gap at current hiring rates |
These numbers define the opportunity structure for AI in African health. The bottleneck is not clinical knowledge — it is the ratio of skilled time to patients.
Accordingly, any tool that enables a community health worker to do what previously required a specialist, or allows a clinician to see 30 patients safely instead of 20, is solving exactly the right problem.
Context: What AI Is Working With in 2026
Before any AI algorithm is evaluated, the infrastructure running it must be considered. A validated tool with a dead tablet battery, a dropped mobile connection, or a generator that ran out of diesel is not helping anyone.
Mobile penetration provides the delivery rail: approximately 80% of adults in sub-Saharan Africa own a mobile phone, and roughly one-third own smartphones. Crucially, digital payment infrastructure is deeply embedded in daily life. Together, these assets make mobile-first health tools feasible in a way that would have been impossible a decade ago.
The constraints are equally real. Only around 28% of the sub-Saharan population regularly accesses the internet. Specifically, fewer than half of rural health facilities have continuous power supply, and only about one-third have broadband access. Infrastructure — not algorithm quality — is typically the binding constraint.
| Rwanda’s solar intervention raised energy uptime in 150 rural facilities from 63% to 98% within one year. That single infrastructure investment turned fragile pilots into reliable services — demonstrating that electricity is itself a medical intervention. |
The training data problem
Developers trained most foundational AI models in use across Africa predominantly on Western datasets. This creates a well-documented performance gap: different skin tone distributions affect dermatology models; different body composition patterns affect chest X-ray interpretation; and different disease prevalence rates change the likelihood ratios underlying clinical decision support.
However, the field is improving. Butterfly Network specifically trained its gestational age AI on data from African populations. Penda Health customised AI Consult with Kenyan epidemiological context and local clinical guidelines. Both represent the right approach — but they remain exceptions rather than norms across the broader tool landscape.
Regulatory landscape: 2025–2026 changes
Notably, the regulatory picture is shifting faster.
- Kenya: Launched its National AI Strategy 2025–2030 in March 2025, allocating KES 152 billion (~$1.14 billion) over five years. The Data Protection Act already governs biometric and health data — Kenya’s High Court enforced it by halting Worldcoin’s iris scan operations.
- Nigeria: The Data Protection Commission introduced enforcement frameworks in September 2025 for the 2023 Data Protection Act. High-risk AI activities including automated decision-making in health now require Data Protection Impact Assessments.
- South Africa: POPIA (Protection of Personal Information Act) anchors AI governance, with the newly launched AI Institute of South Africa (AIISA) developing technical standards for public-sector AI.
- African Union: The Continental AI Strategy (July 2024) provides high-level guidance. The AU’s framework explicitly targets digital sovereignty and African-language AI.
What data localisation means for AI health tools in practice
Of all the near-term regulatory developments, data localisation carries the most direct operational weight. Kenya, Ghana, Nigeria, and Algeria now mandate that certain categories of data be stored or processed within their borders.
For AI health tools that send patient data to foreign cloud servers for processing — which includes most LLM-based clinical decision support — this is not a future compliance risk. It is a current legal requirement in several of the continent’s most active digital health markets. Tool developers and deployers who are not tracking these changes are already exposed.
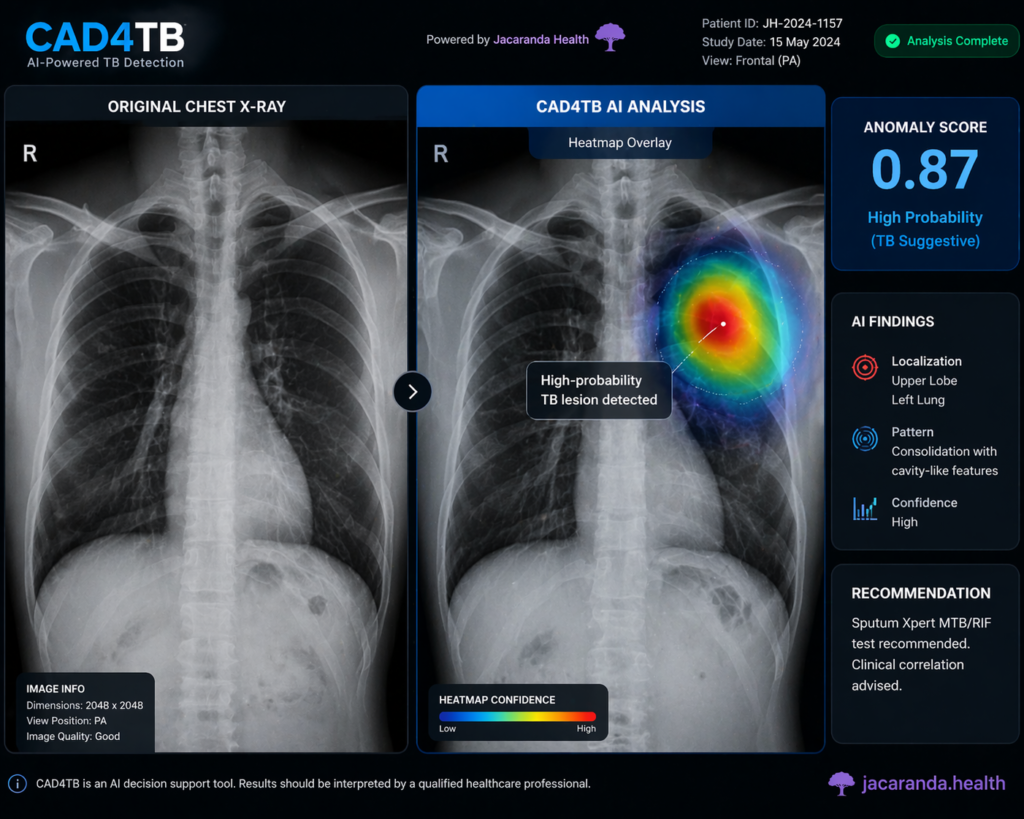
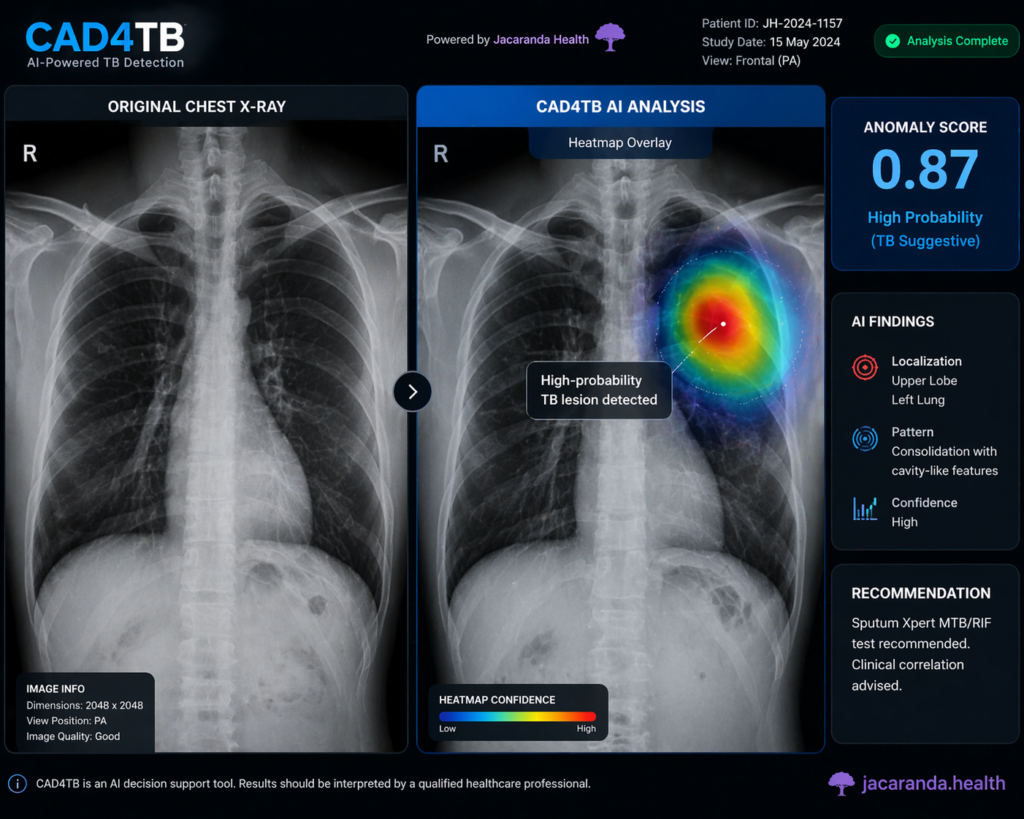
CAD4TB AI software interface showing a chest X-ray heatmap highlighting tuberculosis-suggestive abnormalities with an anomaly score.
AI in TB Diagnostics: The Most Documented Deployment
Tuberculosis remains the world’s leading infectious disease killer, and sub-Saharan Africa carries a disproportionate share of the burden. Consequently, this is — by significant distance — the area where AI health tools in Africa have the most rigorous, most geographically diverse, and most peer-reviewed evidence base.
| CAD4TB has screened over 55 million people in 90+ countries, backed by over 120 peer-reviewed publications — the most validated AI tool for disease detection in any LMIC. |
How AI TB screening works — the triage process
Understanding the tools requires understanding the clinical problem first. TB diagnosis requires microbiological confirmation (GeneXpert or sputum culture) — tests that are expensive and require laboratory infrastructure. AI TB tools do not replace these tests. Instead, they identify who needs them.
The triage process works in four steps:
- X-ray acquisition. An ultra-portable digital X-ray unit (can run on battery power and upload wirelessly) captures a chest radiograph. No fixed infrastructure or mains power required.
- AI analysis. Software such as CAD4TB (Delft Imaging) or qXR (Qure.ai) analyses the image and generates an anomaly score from 0–100, indicating probability of TB-suggestive abnormalities. Results are available within seconds.
- Threshold-based triage. Patients scoring above a pre-set threshold (typically 50 in active case-finding programmes) are flagged as presumptive TB cases and referred for bacteriological testing.
- Bacteriological confirmation. GeneXpert or sputum culture confirms TB and determines drug resistance profile. Treatment begins.
Consequently, the AI’s value sits in steps 2–3: it makes chest X-ray screening operable by non-specialists in the field, enabling genuine active case-finding in communities that would never reach a radiologist.
Evidence: AI vs. standard symptom screening
Multiple head-to-head studies comparing AI-assisted X-ray to the standard WHO four-symptom screening have found consistent results across African settings:
- Sensitivity: AI-assisted X-ray consistently detects more TB cases at equivalent specificity — including asymptomatic cases that symptom screening misses entirely.
- Time to diagnosis: In Nigeria’s national programme, average time to diagnosis fell to just over 2 days, with over 50% of cases diagnosed same-day.
- AUC performance: Leading tools achieve AUC values of 0.87–0.94 in African validation studies, broadly comparable to expert radiologists at matched sensitivity/specificity.
Nigeria: Eight-state rollout with 2026 prison data
Nigeria’s programme is the most evidence-rich multi-context deployment on the continent.
The national programme rolled out 10 ultra-portable X-ray units with CAD4TB across eight states. Published 2025 findings document: average time to diagnosis of just over 2 days; more than 50% same-day diagnosis; 34.5% same-day treatment initiation.
A January 2026 study then extended the evidence to correctional facilities — a high-risk, historically under-screened population. Seventeen adult prisons across 11 Nigerian states were screened. Notably, the study compared CAD4TB directly against the WHO four-symptom screen and found AI-assisted X-ray produced significantly higher TB yield and lower number-needed-to-test.
At the surveillance level
Nigeria piloted CAD4TB+ integrated with the Epi-control predictive analytics platform. Result: predictive mapping guided by the system identified areas with up to 103% higher TB positivity compared to non-targeted screening — demonstrating AI’s potential not just to diagnose, but to direct resources to where they matter most.
Ethiopia: 225 AI units, national rollout
Ethiopia’s deployment is the most ambitious single-country AI health programme in Africa at the time of writing.
Ethiopia procured and distributed 225 AI-powered digital X-ray machines to health facilities across all of the country’s regions for TB screening. To support the rollout, the WHO in Ethiopia trained 75 healthcare workers — including radiology personnel and programme officers — to operate the machines and ensure patients link through to diagnosis and care.
Field evidence from REACH Ethiopia, operating across urban slum, rural, and pastoralist communities, found TB incidence rates of 927 per 100,000 in screened populations — nearly seven times higher than the national estimate. AI-assisted X-ray exhibited significantly higher sensitivity than symptom-based screening, identifying cases that the standard checklist would have missed.
South Africa: Validation data and cost efficiency
South Africa has contributed some of the most rigorous external validation, including use of AI in a national TB prevalence survey — rare population-level evidence. In addition, CAD4TB v7 validation in 1,392 symptomatic adults in South Africa and Lesotho (48% HIV-positive) achieved AUC 0.87 with 68% specificity at 90% sensitivity — comparable to expert radiologist performance and approaching the WHO target product profile.
| Even validated tools go dark when infrastructure fails. Fewer than half of rural sub-Saharan facilities have continuous power, and only one-third have broadband. Algorithm performance is no longer the limiting factor — operational reliability is. |
AI in Maternal Health: 1.8 Million Scans and What They Show
Ninety-two percent of all maternal and neonatal deaths globally occur in low- and middle-income countries. The single largest proximate cause is absence of diagnostic information at the moment a decision needs to be made — too often, clinicians are working without knowledge of gestational age, fetal position, placental location, or early warning signs of pre-eclampsia.
AI is entering this field in two ways: making portable ultrasound interpretable by non-specialists (task-shifting), and adding predictive capability that helps prioritise referrals before complications become emergencies.
Butterfly Network: 1.8 million scans — the numbers behind the milestone
| 1,000 | iQ+ POCUS probes deployed across Kenya and South Africa (Gates Foundation, 2022–2024) |
| 1,050+ | Healthcare providers trained — across nurses, midwives, and community health workers |
| 697+ | Public health facilities with integrated obstetric ultrasound services |
| 1.8M | Scans conducted as of July 2025, averaging 83,000 per month |
| 1.3 min | Average scan time per provider — low enough to be viable in high-volume primary care |
| 99.6% | Provider pass rate on rigorous live clinical assessments post-training |
The preliminary outcome data from South Africa’s Eastern Cape province is the most specific published signal: 20.6% decrease in maternal mortality and 1.13% decrease in stillbirths in the 12 months following training. These are observational findings — other factors may have contributed — but they are consistent with meaningful clinical benefit.
In Kenya, Kenyatta University’s analysis found a significant increase in the volume of women presenting for antenatal care before 24 weeks — an important process outcome, since early presentation is when life-saving interventions can still be timed precisely.
Butterfly Network AI-assisted handheld ultrasound probe conducting a blind sweep for gestational age estimation in Uganda.
The AI gestational age tool: Malawi and Uganda
Launched in October 2025, Butterfly Network’s AI-powered blind-sweep gestational age calculator marked a new frontier for Malawi and Uganda.
Traditional gestational age estimation requires a trained sonographer to identify specific fetal landmarks — a skill that takes months to acquire. Butterfly’s AI tool works differently, through a ‘blind sweep’: the user passes the probe across the abdomen without needing to identify any anatomical structure, and the AI calculates gestational age automatically from the sweep data.
The University of North Carolina developed the tool with Gates Foundation support and — critically — trained it using data from African populations, directly addressing the training data bias problem. Knowing accurate gestational age allows clinicians to time corticosteroids (for prematurity), anti-hypertensives (for pre-eclampsia), and oxytocin (for labour management) precisely rather than by estimation.
Pre-eclampsia prediction: Uganda and Kenya
Predictive algorithms trained on local data are now deployed in Uganda and Kenya to identify women most likely to develop pre-eclampsia. In a reported pilot, referral times for high-risk pregnancies fell by approximately 30%. Meanwhile, in Ghana and Nigeria, AI-supported models are detecting hypertensive disorders and haemorrhage risks in real time during antenatal visits.
These are early-stage deployments. The process metrics are consistent with clinical benefit, but large published RCTs on mortality impact are still forthcoming.
AI in Cancer Screening: Cervical Cancer and Five-Country Evidence
Cervical cancer kills approximately 300,000 women annually. Africa accounts for roughly 23% of global cases. More than 53% of African cases are diagnosed at stage III or IV — advanced stages with poor outcomes. Cervical cancer screening uptake in sub-Saharan Africa sits at approximately 15%, compared to 84% in high-income countries.
AI is entering this field from two angles: improving the accuracy of existing visual screening methods (particularly visual inspection with acetic acid, or VIA), and reducing the specialist training required to perform the initial assessment.
The five-country Lancet AVE study — 2026’s most important new evidence
A prospective diagnostic accuracy study published in The Lancet Global Health in 2026 tested an AI-based Automated Visual Evaluation (AVE) tool in real-world government health facilities across Malawi, Rwanda, Senegal, Zambia, and Zimbabwe.
VIA — the most commonly used cervical screening method in low-resource African settings — has well-documented accuracy limitations that depend heavily on health worker skill and experience. AVE is designed to analyse images of the cervix and flag abnormal findings, assisting health workers in making consistent triage decisions.
The five-country study tested AVE against naked-eye VIA for detection of cervical intraepithelial neoplasia grade 2 or worse (CIN2+). It provides the broadest geographic evidence base yet for AI cervical cancer screening in Africa — and the kind of prospective, multi-country validation that most tools in this field still lack.
Saratani AI: East Africa
Saratani AI is an East Africa-based company operating in Kenya, Nigeria, and Tanzania, developing AI algorithms to analyse PAP smear cytological images for early cervical cancer detection. In October 2025, the company received three Lambda Vector supercomputers donated by Global Health Labs (under the Gates Foundation) to Muhimbili University in Tanzania, dramatically shortening the AI model training cycle from two to three weeks to a matter of days.
The company reports approximately 95% accuracy in pilot testing — a pre-full-validation figure that reflects promising bench results rather than completed clinical validation.
Chil Femtech: Self-collection screening, Uganda and Tanzania
Chil Femtech’s AI-supported self-collection cervical cancer screening platform addresses the most significant cultural barrier to uptake: the social stigma associated with pelvic examination by a clinician. Self-collection allows women to take their own cervical sample; AI analyses the result. This approach has potential to reach populations that conventional clinic-based screening cannot.
LLMs and Clinical Decision Support
The application of large language models to clinical decision support in sub-Saharan Africa is the newest, fastest-moving, and most contested area in the field. It is also — as of 2025 — the area with the most revealing real-world evidence, because two studies of the same class of tool in similar settings produced very different headline conclusions. Understanding why matters for anyone deploying or evaluating LLMs in health.

The Penda Health / OpenAI study
Between January and April 2025, OpenAI and Penda Health — a primary care network operating 16 clinics across Nairobi — conducted what became the largest published real-world evaluation of an LLM clinical decision support tool to date.
| 39,849 | Patient visits across 15 clinics; 20,859 with AI access, 18,990 without |
| 106 | Clinicians randomised between AI-access and non-AI-access groups |
| 5,666 | Visits reviewed by 108 independent physicians (29 from Kenya) |
| -16% | Relative reduction in diagnostic errors in the AI Consult group |
| -13% | Relative reduction in treatment errors in the AI Consult group |
| -31% | Diagnostic error reduction in the highest-risk encounters (red-alert cases) |
| 75% | Share of AI-group clinicians saying the tool had a ‘substantial’ effect on their care quality |
The tool — AI Consult, built on GPT-4o — ran silently in the background during every patient visit, activating only when it detected a potential error. It displayed a traffic-light alert (green / yellow / red) and offered specific recommendations. Critically, it was customised for Kenyan clinical practice: prompts included local disease prevalence data, national guidelines, and Penda’s standard procedures.
An unexpected secondary finding: clinicians in the AI group triggered fewer red alerts over time — evidence of a learning effect, with the AI improving clinician reasoning even before its feedback was delivered. This is one of the most significant implications of the study and one of its least-discussed findings.
| Penda is now running a full randomised controlled trial with PATH to measure patient outcomes directly — not just documentation quality. Those results will be the definitive test of whether the error reductions translate to lives saved. |
The ‘safety gap’: a counter-narrative from Nature Health
Published in March 2026 in Nature Health, a separate retrospective safety evaluation of an LLM clinical decision support system deployed across 16 primary care clinics in Kenya — reviewing 1,469 patient records — produced findings that complicate the Penda narrative.
However, one number received far less media coverage than it deserved:
Physicians independently identified actively harmful recommendations in 7.8% of encounters (115 total), with 67 of those appearing in the final clinical documentation. Furthermore, clinicians did not modify documentation in 62% of encounters — suggesting frequent non-engagement with AI output, whether positive or negative.
To be fair, the reassuring findings matter too: clinical guidance aligned with local guidelines in 99% of cases, and hallucinations remained uncommon at 3.4%, most often involving misexpanded acronyms or drug names.
| These findings are not contradictory to the Penda results — they reflect different implementation quality. Penda invested in one-on-one clinician coaching, peer champions, performance feedback, and leadership recognition. Consequently, the unresolved red-alert rate fell from 40% to 20% over the study period. By comparison, the Nature Health study does not describe equivalent implementation support in the control group. Implementation rigour is therefore as important as algorithm quality. |
| An AI recommendation that is systematically ignored is not improving care. An AI recommendation that is harmful and also ignored may be worse than no AI at all. |
Speech-to-text AI: 48 hours to 20 minutes, UCH Ibadan
Dr Tobi Olatunji’s clinical speech-to-text AI — the first trained specifically on African accents — demonstrates a simpler, more immediately beneficial application at University College Hospital Ibadan. Radiology reporting time fell from 48 hours to 20 minutes. Doctors dictate; the AI transcribes and structures. Crucially, no diagnostic uncertainty enters the workflow, and the change eliminates administrative friction entirely.
This category of application — workflow automation rather than clinical decision-making — carries substantially lower risk and delivers immediate measurable benefit. It may be underinvested relative to the harder diagnostic AI problems.
Rwanda’s National Health Intelligence Centre
Rwanda has taken the most nationally integrated approach to AI in health: building the data infrastructure before deploying point-of-care tools. The National Health Intelligence Centre consolidates data from multiple health system sources and applies AI analytics for real-time surveillance and early outbreak detection. This system-level investment means that when clinical tools are deployed, they operate within a data ecosystem that can measure their impact — not in a vacuum.
AI in the Supply Chain: The Missing Link Between Diagnosis and Treatment
So far, the diagnostic AI field has focused intensely on finding disease earlier. Yet the question that immediately follows is: after a correct AI-assisted diagnosis, can the patient actually get the right medicine? In sub-Saharan Africa, the answer is frequently no — not because the diagnosis was wrong, but because the medicine is not in stock.
Supply chain AI closes this loop. Two platforms operating at scale illustrate how.
Zipline: AI logistics at continental scale
Zipline is an autonomous drone delivery company that has been operating in African health systems since 2016. By early 2026, it had completed its two millionth commercial delivery, was making a delivery every 60 seconds, and was serving over 4,800 health facilities across Rwanda, Ghana, Nigeria, Kenya, and Côte d’Ivoire — reaching approximately 49 million people.
Zipline’s operations are not simply ‘drones with packages.’ The underlying logistics intelligence — route optimisation, demand forecasting, inventory management, and emergency response prioritisation — is AI-powered. The Rwanda Lancet study found the service shortened blood product delivery times by 61% and reduced blood unit expirations by 67%, with 43% of orders classified as emergency orders.
An independent Wharton study (not peer-reviewed) reported a 51% reduction in deaths from postpartum haemorrhage in Rwanda attributable to Zipline-delivered blood.
In Ghana, an independent study similarly found that Zipline reduced vaccine stockouts by 60% and decreased inventory-driven missed vaccination opportunities by 42%.
The November 2025 expansion deal
The US State Department committed up to $150 million to expand Zipline’s network from 5,000 to 15,000 health facilities across Rwanda, Kenya, Nigeria, Ghana, and Côte d’Ivoire — potentially reaching 130 million additional people. African governments committed to approximately $400 million in utilisation fees as part of a pay-for-performance structure.
| Important nuance from a February 2026 study of Zipline’s Ghana operations: a majority of healthcare workers perceived improvements in emergency supply delivery (50–54%) but fewer observed improvements in routine service delivery (29%). Drones are most impactful in emergency logistics — their value for routine supply chain management is less established. |
Maisha Meds: AI-powered pharmacy networks, Kenya to Nigeria
While Zipline handles last-mile delivery, a complementary platform addresses the stock management and prescribing intelligence layer at the pharmacy and clinic level.
Founded in 2017, Maisha Meds now operates the largest digital network of private pharmacies and clinics in Africa: approximately 4,600 drug shops, pharmacies, and clinics across Kenya, Uganda, Tanzania, Nigeria, and Zambia, logging over 25 million patient visits annually. The platform is expanding toward 7,500 facilities by late 2026.
Within the same app providers use to run their businesses, Maisha Meds has embedded AI-enabled features: stock-out and expiry forecasting, demand prediction for seasonal and outbreak-driven surges, and claims auditing powered by AI to flag inappropriate prescribing and ensure subsidy payments go to compliant care.
Prescribing quality
Unusually for this category, the evidence for prescribing quality impact is rigorous: a randomised controlled trial with UC Berkeley found that Maisha Meds’ reimbursement programme — which uses AI-enabled auditing to verify appropriate care — more than quadrupled provider adherence to malaria testing and treatment protocols in Kenya, increasing compliance from 8% to 33%.
As a result, combining diagnostic AI (catching the disease) with supply chain AI (ensuring the medicine is in stock) and pharmacy AI (verifying appropriate prescribing and dispensing) creates a closed loop — the full care pathway, not just the detection endpoint.
AI and Mental Health: The Highest-Need, Lowest-Evidence Area
Sub-Saharan Africa has a mental health care deficit that dwarfs even the physical health worker shortage. The continent accounts for a disproportionate share of global mental illness burden, yet psychiatric services are even thinner on the ground than general medicine: in many countries, there is a single psychiatrist per million people.
AI chatbots and digital mental health tools are entering this space rapidly — in some cases faster than they are being evaluated. The evidence base globally for AI mental health tools is evolving, and Africa-specific evidence is particularly sparse.
What the global evidence shows
A 2025 systematic review of 160 mental health chatbot studies (2020–2024) found that LLM-based chatbots surged to 45% of new studies in 2024 — but only 16% of those LLM studies underwent clinical efficacy testing.
Furthermore, a meta-analysis across 15 RCTs found AI-based conversational agents significantly reduced symptoms of depression (Hedge’s g 0.64) and psychological distress (Hedge’s g 0.70). These are meaningful effect sizes — comparable to low-intensity psychological interventions.
The limitations are well-documented: low user retention, cultural sensitivity gaps, algorithmic bias, and the specific risks of deploying emotional support AI in crisis situations without clear escalation pathways.
Africa-specific deployments
Several AI-assisted mental health platforms are operating in urban hubs across sub-Saharan Africa, primarily targeting populations where psychiatrist access is near-zero:
- Zuri Health (Kenya): AI-powered triage and mental health counselling platform handling significant volumes of queries in urban Kenya. The platform routes users to appropriate human support when AI triage flags clinical need — a hybrid model that represents best practice for this application category.
- WhatsApp-based mental health bots: Several platforms across West Africa are using WhatsApp — the continent’s dominant messaging platform — to deliver structured psychoeducation and basic psychological first aid, leveraging existing user behaviour rather than requiring new app adoption.
Honestly, the need is enormous, the tools are operating, and the Africa-specific evidence is thin. Mental health AI in Africa is closer to the ‘promising but early’ tier than any other category in this article. That said, the risks — unsafe escalation in crisis, cultural mismatch, and over-reliance — are real and require governance frameworks that most operating platforms do not yet have.
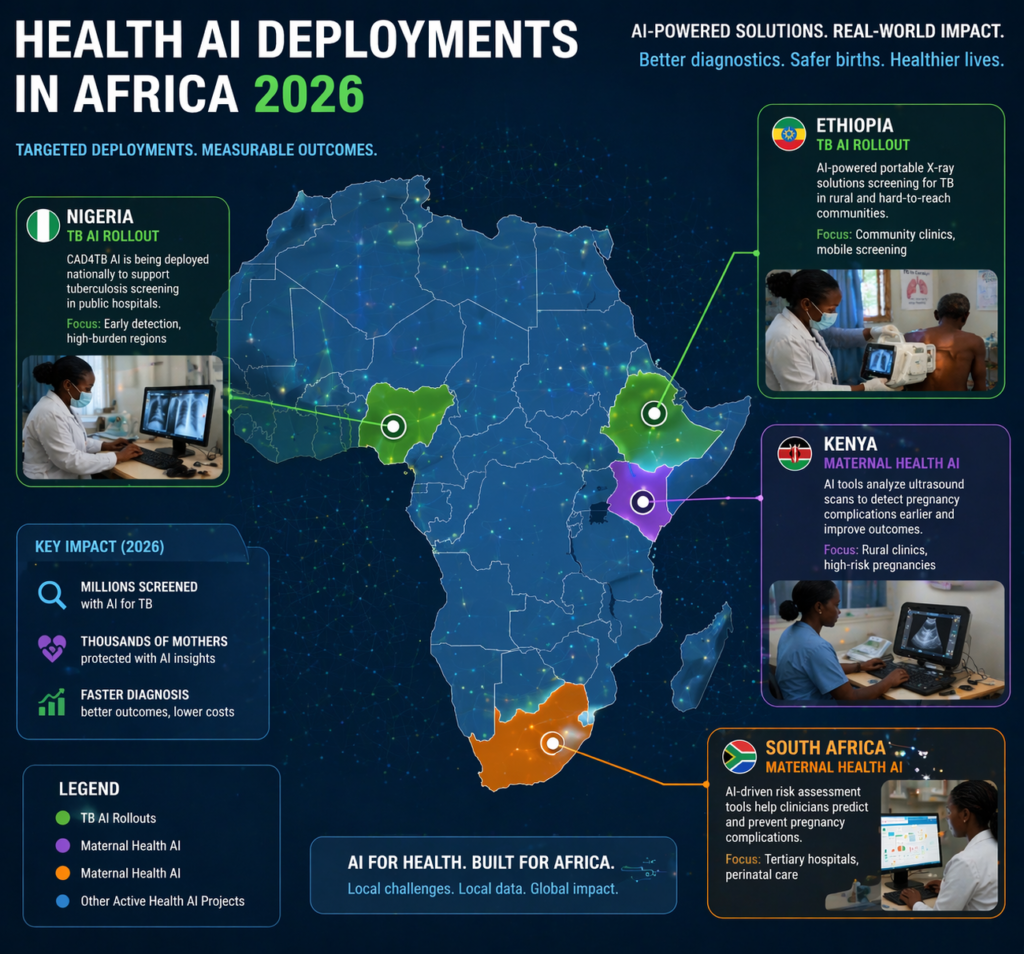
Map of health AI deployments in Africa 2026 showing national TB rollouts in Ethiopia and Nigeria and maternal health AI in Kenya and South Africa.
The maturing cohort of African-founded startups is a direct result of the evolving AI ecosystem across the continent, where local ownership of code is becoming a strategic priority
Country-by-Country Snapshot
The table below maps documented AI health deployments to specific countries. Only tools with verifiable scale indicators or published evidence are included.
| Country | Deployed Tools | Scale Indicator | Key Evidence / Status |
| Ethiopia | 225 AI X-ray units (CAD4TB); national TB programme | National rollout, all regions | 7× higher TB incidence found vs. national estimate; WHO-supported training |
| Kenya | AI Consult (Penda/OpenAI); Butterfly POCUS; CAD4TB; Maisha Meds | 16 clinics; 697+ POCUS facilities; 4,600+ pharmacy network | 39,849-patient study; 16% diagnostic error reduction; 1.8M scans; RCT malaria compliance +25pp |
| Nigeria | CAD4TB (8 states); speech-to-text (UCH Ibadan); Maisha Meds; Zipline | National TB; 4,600+ pharmacies; drone in 3 states | 103% TB positivity in targeted areas; reporting time 48h → 20 min |
| Rwanda | National Health Intel Centre; Zipline; Horizon1000 (launching) | National data integration; every 60s delivery | Energy uptime 63%→98% (solar); blood delivery time –61%; $50M committed |
| Uganda | AI obstetric ultrasound (Makerere); Butterfly POCUS; Chil Femtech | Community health worker level | Enabling non-specialists to conduct fetal imaging |
| South Africa | CAD4TB (prevalence surveys); Butterfly POCUS; Envisionit Deep AI; Zipline | 697+ POCUS facilities (with Kenya); national TB survey | Cost per TB case $1,748→$437; 20.6% maternal mortality decrease (Eastern Cape) |
| Ghana | minoHealth AI Labs (chest X-ray); Zipline | West Africa AI hub; drone in 6 centres | AUC-ROC 0.97 X-ray AI; vaccine stockouts –60% |
| Malawi | Butterfly Gestational Age AI; AVE cervical screening study | Facility level (launched Oct 2025) | Part of Lancet Global Health 5-country AVE study |
| Tanzania | Saratani AI (clinical validation); Chil Femtech; Maisha Meds | Clinical validation ongoing; pharmacy network | 95% accuracy claim (pre-validation); Gates computers donated to MUHAS |
| Zambia | CAD4TB pilots; AVE cervical study; diabetic retinopathy AI; Maisha Meds | Research/early scale | Clinically acceptable DR AI performance; pharmacy AI network |
| Zimbabwe | AVE cervical screening study | Research deployment | Lancet Global Health 5-country study |
| Lesotho | CAD4TB (validation study) | Research/programme support | CAD4TB v7 AUC 0.87 in HIV-positive population |
| Senegal | AVE cervical screening study | Research deployment | Lancet Global Health 5-country study |
| Côte d’Ivoire | Zipline AI drone delivery | National programme | Part of US State Dept $150M expansion deal (Nov 2025) |
What the Evidence Actually Shows: An Honest Tier Assessment
Above all, the most useful thing this article can do is distinguish what has been proven from what has been deployed and what has merely been announced. Each tier below reflects the depth and rigour of available evidence, not the ambition of the tools.
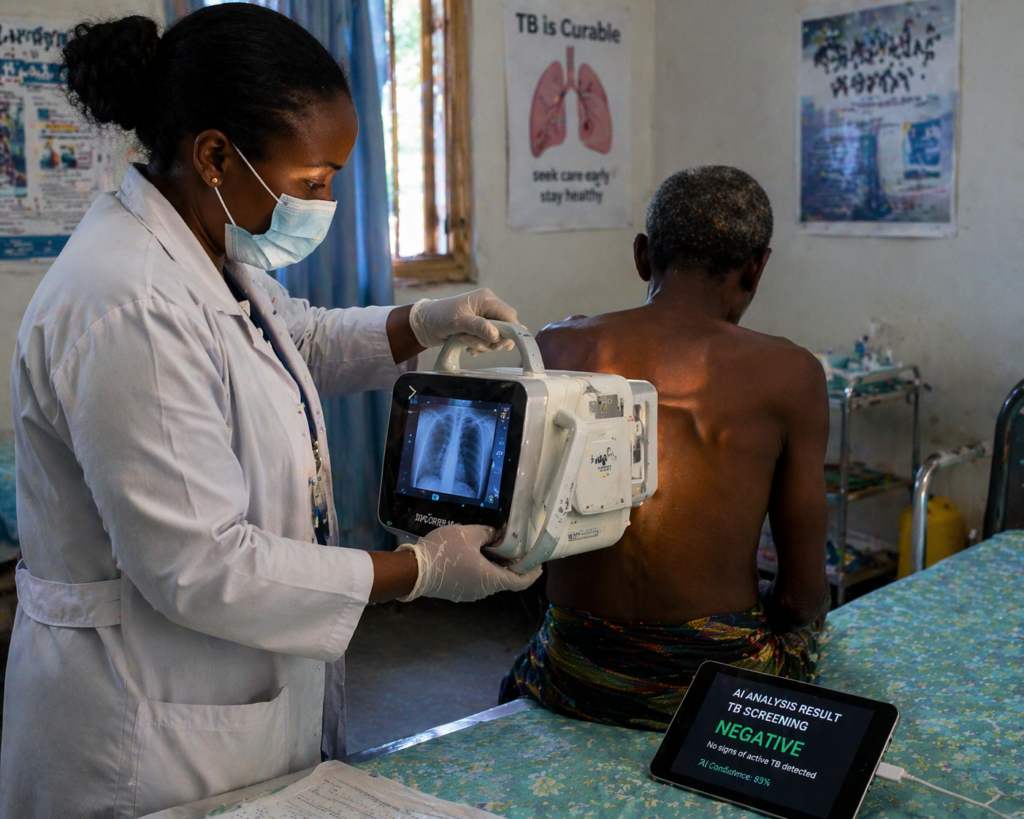
Healthcare worker in a rural South African clinic using an AI-powered portable X-ray machine for tuberculosis screening in 2026.
Tier 1 — : TB diagnostics and supply chain AI
CAD4TB and comparable AI chest X-ray tools have the deepest, most geographically diverse evidence base in African health AI: 120+ peer-reviewed publications, 55M+ people screened, external validation in multiple African settings with consistent performance at or near expert radiologist level.
The cost-efficiency data from South Africa ($437 vs $1,748 per case found) is real-world programme evidence, not a modelled projection. Ethiopia’s national deployment and Nigeria’s correctional facility data are 2025–2026 evidence, not legacy.
Zipline’s supply chain AI is similarly well-evidenced: the Rwanda Lancet study (61% faster blood delivery, 67% fewer expirations) and Ghana independent study (60% fewer vaccine stockouts) represent the kind of before-after programme data that health economists can act on. The Maisha Meds UC Berkeley RCT (malaria protocol adherence: 8% → 33%) is the most rigorously designed evaluation of pharmacy AI impact in Africa.
Tier 2 — : Maternal health AI and LLM decision support
Butterfly’s 1.8M scans and the Eastern Cape outcome data (20.6% maternal mortality reduction, 1.13% stillbirth reduction) are consistent with meaningful clinical benefit. That said, the caveat is honest: these are observational findings, and the randomised controlled trial is in progress.
Why the two Kenya LLM studies are not contradictory
The Penda/OpenAI study is genuine real-world evidence: 39,849 visits constitutes a large sample, and the 16% relative error reduction is meaningful. However, it comes from one network of clinics in one city with an unusually well-resourced implementation programme. Generalising those results to the African primary care median would therefore be premature.
Tier 3 —: Cervical cancer screening and mental health AI
The five-country Lancet Global Health AVE study is the most important piece of new cervical cancer evidence — and it is very new. Saratani AI is in clinical validation. Mental health chatbots are operating in African urban settings but with minimal Africa-specific efficacy evidence.
The publication bias problem
| The published evidence skews toward successful deployments in Kenya, South Africa, Nigeria, and Ethiopia — and toward TB, HIV, and maternal health. Tools that failed, pilots that went dark when donor funding ended, and algorithms that performed poorly in local validation are largely unpublished. The real evidence base is stronger than five years ago, but it is not as strong as the literature alone suggests. |
The Real Barriers to Scale in 2026
In sub-Saharan Africa, the barriers to AI health scale are not primarily technical. Algorithms exist that work at or near specialist level for specific tasks, and teams have deployed them. Instead, what holds back scale is systemic, structural, and human.
1. Power and connectivity: still the binding constraint
Even validated, proven tools fail when the generator runs out. A Ghanaian digital triage system that reduced referral delays by 30% went silent when donor funding ended and facilities could not afford generator fuel. Rwanda’s solar infrastructure investment is the most important case study: it did not require novel technology — it required political will and sustained funding.
2. Training data bias: narrowing but not closed
Developers built most foundational AI models now in use across Africa using data from elsewhere. As a result, systematic performance gaps arise in radiology, dermatology, and clinical text processing — gaps the literature documents consistently. The gap is narrowing — Butterfly’s gestational age AI, Penda’s LLM customisation — but it remains open for the majority of tools currently in the field.
3. Data sovereignty: the 2026 regulatory shift
This is the most rapidly evolving barrier. Data localisation laws are tightening across the continent:
- Kenya’s Cloud Policy (May 2025): Mandates data localisation by classification level. Top Secret and Secret government data must be hosted in Kenyan government-cloud infrastructure.
- Nigeria’s NDPA enforcement (Sept 2025): High-risk AI activities in health now require Data Protection Impact Assessments and certified Data Protection Officers.
- Kenya High Court (late 2025): Issued conservatory orders suspending data transfer provisions of a USD 1.6 billion US Health Cooperation Framework, ruling that sharing seven years of epidemiological data lacked adequate safeguards.
For AI tools that process patient data through foreign cloud servers — which includes most LLM-based clinical decision support — this regulatory shift already demands compliance in several of the continent’s most active digital health markets. Tool developers and deployers who are not tracking these changes are therefore exposed now, not in the future.
As of mid-2025, Africa has 223 data centres across 38 countries, led by South Africa (56 facilities), Kenya (19), and Nigeria (17). Driven partly by AI adoption and partly by local data residency requirements, the data centre market is projected to reach $5 billion by 2026.
Why non-engagement with AI output is as dangerous as harmful output
Kenya’s LLM evidence offers the clearest statement of the workflow trust problem: clinicians did not modify documentation in 62% of encounters in the Nature Health study. An AI recommendation that is routinely ignored has zero clinical impact — and where that recommendation was harmful and still appeared in the record, it may have introduced legal liability.
Penda fixed this by investing in sustained implementation support — one-on-one coaching, peer champions, and performance feedback. That lesson is transferable but costly: real adoption requires ongoing investment in training and change management, not a one-off onboarding session.
5. Sustainability after donor funding
Across sub-Saharan Africa, the modal fate of health AI pilots is collapse when the grant cycle ends. By contrast, the tools now scaling successfully — Butterfly, Ethiopia’s national TB programme, Penda, Maisha Meds, Zipline — share a common feature: they are embedded in national programmes or sustainable business models, not dependent on a single donor.
6. The ‘built elsewhere’ problem and the African AI response
Teams outside Africa built the majority of currently deployed tools. That is changing — minoHealth (Ghana), Saratani AI (East Africa), Neural Labs Africa (Kenya), Chil Femtech (Uganda) — yet the dependency remains significant. By contrast, locally built tools carry stronger incentives to incorporate local clinical context and stronger reasons to remain operational when international partners eventually move on.
The 2028 Roadmap: From Scale to Sovereignty
The trajectory from 2026 to 2030 is now more defined than at any previous moment. AI will clearly be part of African health systems — it already is. What remains open is whether deployment will be safe, locally owned, and structured so that the value stays on the continent.
Horizon1000: The biggest active commitment
In January 2026, the Gates Foundation and OpenAI committed $50 million to Horizon1000 — equipping 1,000 primary care clinics across sub-Saharan Africa with AI tools by 2028, starting in Rwanda. Both organisations frame it explicitly as a leapfrog: Rwanda’s 180-year timeline to close its health worker gap at current hiring rates is not a planning horizon. Horizon1000 is designed to circumvent it.
Nevertheless, the success conditions are well-understood from the evidence: context-specific customisation, sustained implementation support, functional infrastructure, and local ownership of both data and tools. Whether Horizon1000 delivers on all four will be the defining evaluation of the initiative.

A Solar-powered rural health facility in Rwanda provides continuous electricity for AI-driven diagnostic tools and digital health records.
Zipline’s expansion: 5,000 to 15,000 facilities
The US State Department’s $150 million commitment to expand Zipline’s network across Rwanda, Kenya, Nigeria, Ghana, and Côte d’Ivoire — with African governments committing ~$400 million in utilisation fees under a pay-for-performance structure — is the largest single investment in AI health logistics on the continent. Reaching up to 130 million additional people, and tripling the facility count from 5,000 to 15,000 by ~2028.
The data sovereignty transition: from compliance to competitive advantage
The regulatory changes described in Section 11 are reframing data sovereignty from a compliance burden to a strategic asset. Countries and companies that invest in local data infrastructure, local AI training, and regulatory frameworks that protect patient data are building competitive moats. Those that don’t are building liability.
The Microsoft-G42 $1 billion geothermal-powered data centre in Kenya’s Naivasha zone (operational by 2026) is the clearest signal that hyperscalers are responding to local data residency requirements — not just waiting for African markets to conform to global infrastructure models.
What success looks like in 2030
Consider what concrete success looks like. A child born near Kisumu or Kumasi should not need to travel 200 kilometres to find out whether a cough is tuberculosis or a cervical lesion is cancerous. Encouragingly, the tools to make that possible are either already deployed or in late-stage validation.
What still needs to happen — in parallel, not sequentially: infrastructure investment (solar, connectivity) that makes tools reliably usable; data sovereignty frameworks that keep the value of African health data in African hands; implementation support that is sustained, not one-off; and genuine local ownership of both the algorithms and the decisions about how they shape care.
A first wave of genuinely scaled, evidence-based AI health tools in sub-Saharan Africa is already real. Moreover, a second wave — broader, deeper, locally owned, and grounded in regulation that protects patients — is the one that will define the decade.
Frequently Asked Questions
What AI health tools are currently deployed at scale in sub-Saharan Africa?
As of early 2026, several tools have moved beyond the pilot phase into verifiable scale:
- TB Screening: CAD4TB (225 units in Ethiopia; 8-state rollout in Nigeria; national survey use in South Africa).
- Maternal Health: Butterfly Network POCUS (1.8M scans across 697+ facilities in Kenya and South Africa).
- Clinical Support: AI Consult LLM (active across all 16 Penda Health clinics in Nairobi).
- Cervical Cancer: AVE Screening (validated across Malawi, Rwanda, Senegal, Zambia, and Zimbabwe).
- Logistics & Pharmacy: Zipline AI (4,800+ facilities) and Maisha Meds (4,600+ sites, 25M annual patient visits).
Is AI healthcare cost-effective in sub-Saharan Africa?
Yes. Data-driven outreach using the CAD4TB+ platform in South Africa reduced the cost per TB case found from $1,748 to $437—a 75% reduction. In Nigeria’s UCH Ibadan, AI speech-to-text reduced radiology reporting time from 48 hours to 20 minutes, dramatically increasing patient throughput without the need for additional specialized staff.
Is AI replacing doctors in Africa?
No. The dominant model is task-shifting. AI tools extend the capabilities of community health workers, nurses, and midwives to perform tasks that previously required specialists. For example, Butterfly’s gestational age tool allows midwives to conduct fetal imaging with no prior interpretation training. The bottleneck being solved is the ratio of skilled clinician time to patients, not the replacement of clinical judgment.
What is the Horizon1000 initiative?
Horizon1000 is a joint initiative launched in January 2026 by the Bill & Melinda Gates Foundation and OpenAI. It commits $50 million to equip 1,000 primary healthcare clinics across sub-Saharan Africa with AI tools by 2028. The program begins in Rwanda, acting as a “leapfrog” strategy to address a health worker shortfall that would otherwise take 180 years to close at current hiring rates.
What do the LLM clinical decision support studies in Kenya actually show?
The evidence is nuanced and should be read as a pair:
- The Penda Health/OpenAI Study (2025): Evaluated 39,849 visits and found a 16% reduction in diagnostic errors and a 13% reduction in treatment errors.
- The Nature Health Safety Study (2026): Found 99% alignment with local guidelines but identified potentially harmful recommendations in 7.8% of encounters.
- The Verdict: The difference reflects implementation quality. Penda invested heavily in clinician training and peer champions, whereas the second study highlights that AI deployment without sustained human support is insufficient and potentially risky.
How is Africa addressing data sovereignty for AI health tools?
Regulation is rapidly tightening. Kenya’s Cloud Policy (May 2025) and Nigeria’s Data Protection Commission frameworks now mandate strict data localization for high-risk AI activities. Notably, in late 2025, Kenya’s High Court suspended data transfer provisions of a $1.6 billion US health deal, ruling that sharing years of epidemiological data lacked adequate safeguards. AI tools using foreign cloud servers now face significant compliance exposure in Kenya, Ghana, Nigeria, and Algeria.
Are there any African-built AI health tools?
Yes. A maturing cohort of African-led companies is building tools specifically for local clinical contexts:
- minoHealth AI Labs (Ghana): Diagnostic AI for chest X-rays.
- Saratani AI (Tanzania): Cervical cancer screening.
- Neural Labs Africa (Kenya): Real-time medical imaging.
- Chil Femtech (Uganda): Tele-oncology and self-collection screening.
What are the main barriers to scaling AI health tools in Africa?
The evidence identifies five primary systemic barriers:
- Infrastructure: Fewer than 50% of rural facilities have continuous power; however, Rwanda’s solar interventions (raising uptime from 63% to 98%) prove this is solvable.
- Training Data Bias: Models built on Western datasets often underperform on African populations.
- Regulatory Gaps: While improving in 2026, many nations still lack specific AI safety frameworks.
- Workflow Integration: Without proper training, up to 62% of AI-generated insights go unactioned by clinicians.
- Sustainability: Many advanced tools remain dependent on donor funding and struggle to survive once grant cycles end.
Related Reading: The Rise of AI in Africa: How Digital Infrastructure is Shaping the Future